5-APDI
 | |
| Clinical data | |
|---|---|
| Other names | 5-APDI; 1-(5-Indanyl)-2-aminopropane; Indanylaminopropane; IAP; Indanametamine; 2-Aminopropylindane; 2-API; Indanylamphetamine |
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
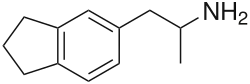
| Formula | C12H17N |
| Molar mass | 175.275 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
SMILES
| |
InChI
| |
| (verify) | |
5-(2-Aminopropyl)-2,3-dihydro-1H-indene (5-APDI), also known as indanylaminopropane (IAP), 2-aminopropylindane (2-API), indanametamine, and, incorrectly, as indanylamphetamine,[1] is an entactogen and psychedelic drug of the amphetamine family.[2][3] It has been sold by online vendors through the Internet and has been encountered as a designer drug since 2003,[1] but its popularity and availability has diminished in recent years.
5-APDI appears to act as a potent and weakly selective serotonin releasing agent (SSRA) with IC50 values of 82 nM, 1,848 nM, and 849 nM for inhibiting the reuptake of serotonin, dopamine, and norepinephrine, respectively.[2][3] It fully substitutes for MBDB but not amphetamine in trained animals, though it does produce disruption for the latter at high doses.[2]
5-APDI has been classified as a class B drug under the Misuse of Drugs Act 1971 since 10 June 2014.
In a 1968 patent, 5-APDI was fused with 3,3-Diphenylpropylamine.[4] The dose (of the HCl salt) was judged to be 55mg per tablet (corr. to 50mg of the Fb). The compounds has valuable pharmacodynamic properties whilst its toxicity is low. It produces a vasodilatation and thus improve peripheral blood circulation and a pronounced coronary dilatation. The compound further exhibits a blood pressure lowering effect and is therefore indicated for use in the treatment of hypertonia and circulatory illnesses, especially Angina pectoris and other stenocardiac disorders and in the treatment of organic or functional coronary insufficiencies and peripheral blood circulation disorders.
See also
References
- 1 2 Casale JF, McKibben TD, Bozenko JS, Hays PA (2005). "Characterization of the "Indanylamphetamines"". Microgram Journal. 3 (1–2): 3–10. Archived from the original on 2009-03-17. Retrieved 2009-08-06.
- 1 2 3 Monte AP, Marona-Lewicka D, Cozzi NV, Nichols DE (November 1993). "Synthesis and pharmacological examination of benzofuran, indan, and tetralin analogues of 3,4-(methylenedioxy)amphetamine". Journal of Medicinal Chemistry. 36 (23): 3700–6. doi:10.1021/jm00075a027. PMID 8246240.
- 1 2 Parker MA, Marona-Lewicka D, Kurrasch D, Shulgin AT, Nichols DE (March 1998). "Synthesis and pharmacological evaluation of ring-methylated derivatives of 3,4-(methylenedioxy)amphetamine (MDA)". Journal of Medicinal Chemistry. 41 (6): 1001–5. CiteSeerX 10.1.1.688.9559. doi:10.1021/jm9705925. PMID 9526575.
- ↑ Frank Troxler & Albert Hofman, GB1133457 (1968 to Sandoz KK).
External links
| DRAsTooltip Dopamine releasing agents |
| ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NRAsTooltip Norepinephrine releasing agents |
| ||||||||||||||
| SRAsTooltip Serotonin releasing agents |
| ||||||||||||||
| Others |
| ||||||||||||||
See also: Receptor/signaling modulators • Monoamine reuptake inhibitors • Adrenergics • Dopaminergics • Serotonergics • Monoamine metabolism modulators • Monoamine neurotoxins | |||||||||||||||
| Phenethylamines |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amphetamines |
| ||||||||||||||||
| Phentermines |
| ||||||||||||||||
| Cathinones |
| ||||||||||||||||
| Phenylisobutylamines (and further-extended) | |||||||||||||||||
| Catecholamines (and close relatives) |
| ||||||||||||||||
| Cyclized phenethylamines |
| ||||||||||||||||
| Related compounds |
| ||||||||||||||||
| |||||||||||||||||
Chemical classes of psychoactive drugs | |
|---|---|
| Stimulants |
|
| Depressants |
|
| Hallucinogens |
|
| Entactogens |
|
| Psychiatric drugs |
|
| Others |
|